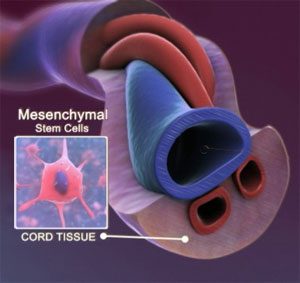
CytoCord™
Login to see pricesUmbilical Cord Matrix/Wharton’s Jelly Nucleated Native product contains both growth factors and an assortment of cell types, including mesenchymal stem cells, epithelial cells, and endothelial cells. These cell types can secrete anti-inflammatory and immunomodulatory properties that can then be optimized to reach your clinical research needs.
All donors go through a rigorous screening process, as determined by the guidelines and regulations listed by the FDA and American Association of Tissue Banks (AATB). Maternal blood for the fresh umbilical cord tissue must be negative for HIV (Human Immunodeficiency Virus), HBV (Hepatitis B), HCV (Hepatitis C), CMV IgM (cytomegalovirus antibodies), Syphilis, WNV (West Nile Virus), HTLV I/II (Human T-Cell Lymphotropic Virus Types I and II), and Chagas Disease. The consented umbilical cord ships to our cell manufacturing facility where primary cells are processed and analyzed. Parameters including cell phenotype, viability, purity, and cell count are checked to meet the FDA’s gold standard for biopharma companies, ensuring high-quality cells are characterized and purified.